UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM
CURRENT REPORT
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
(Exact name of Registrant as Specified in Its Charter)
| (State or Other Jurisdiction of Incorporation) |
(Commission File Number) |
(IRS Employer Identification No.) |
| (Address of Principal Executive Offices) | (Zip Code) |
Registrant’s Telephone Number, Including Area Code:
(Former Name or Former Address, if Changed Since Last Report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class |
Trading |
Name of each exchange | ||
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
| Item 7.01 | Regulation FD Disclosure. |
On September 8, 2025, Dianthus Therapeutics, Inc. (the “Company”) issued a press release announcing positive top-line data from the Phase 2 MaGic trial evaluating the safety and efficacy of claseprubart (DNTH103) in adults with acetylcholine receptor antibody positive (AChR+) generalized Myasthenia Gravis (gMG). The Company will host a conference call and webcast today, Monday, September 8, 2025, at 8:00 am, Eastern Time, to discuss the data results.
A copy of the press release is furnished as Exhibit 99.1 to this Current Report on Form 8-K and is incorporated by reference herein. The exhibit furnished under Item 7.01 of this Current Report on Form 8-K shall not be deemed to be “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Exchange Act or the Securities Act of 1933, as amended, regardless of any general incorporation language in such filing.
| Item 8.01 | Other Events. |
On September 8, 2025, the Company made publicly available a data presentation announcing positive top-line data from the Phase 2 MaGic trial evaluating the safety and efficacy of claseprubart (DNTH103) in adults with acetylcholine receptor antibody positive (AChR+) generalized Myasthenia Gravis (gMG) (the “Presentation”) on the investor relations section of its website. The Presentation is filed as Exhibit 99.2 and is incorporated by reference into this Item 8.01.
| Item 9.01 | Financial Statements and Exhibits. |
(d) Exhibits. The following exhibit is being furnished herewith:
EXHIBIT INDEX
| Exhibit No. |
Description | |
| 99.1 | Data Press Release, dated September 8, 2025 | |
| 99.2 | Data Presentation, dated September 8, 2025 | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document). | |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| DIANTHUS THERAPEUTICS, INC. | ||||||
| Date: September 8, 2025 | By: | /s/ Adam M. Veness, Esq. | ||||
| Adam M. Veness, Esq. | ||||||
| SVP, General Counsel and Secretary | ||||||
Exhibit 99.1

|

|
DIANTHUS THERAPEUTICS ANNOUNCES POSITIVE DATA FOR CLASEPRUBART (DNTH103) FROM THE PHASE 2 MAGIC TRIAL IN GENERALIZED MYASTHENIA GRAVIS, SUPPORTING ITS POTENTIAL BEST-IN-CLASS PROFILE
Claseprubart 300mg and 600mg Q2W doses both achieved statistically significant and clinically meaningful improvements in Myasthenia Gravis Activities of Daily Living (MG-ADL) and Quantitative Myasthenia Gravis (QMG) scores at Week 13
Claseprubart demonstrated a rapid and sustained onset of action, with both doses achieving statistically significant improvements in MG-ADL and QMG scores at Week 1
Claseprubart 300mg Q2W dose was also statistically significant and clinically meaningful across other key efficacy endpoints, including Minimal Symptom Expression (MSE), Myasthenia Gravis Composite (MGC) Score and the Myasthenia Gravis Quality of Life Scale (MG-QoL-15r)
Both 300mg and 600mg doses of claseprubart were comparable across key efficacy endpoints, supporting its target product profile of a single, convenient 300mg/2mL self-administered, subcutaneous autoinjector dosed once every two weeks
Claseprubart had a favorable safety profile with no related serious infections, clinical symptoms of emergent autoimmune disease, or drug-related serious adverse events or discontinuations, supporting its target product profile of no Boxed Warning or REMS for meningococcal infections
Phase 3 gMG trial evaluating 300mg/2mL Q2W and Q4W vs. placebo anticipated to initiate in 2026
Investor conference call and webcast to be held today, September 8, 2025 at 8:00 a.m. ET
New York City and Waltham, Mass., September 8, 2025 – Dianthus Therapeutics, Inc. (Nasdaq: DNTH), a clinical-stage biotechnology company dedicated to advancing the next generation of antibody complement therapeutics to treat severe autoimmune diseases, today announced positive top-line data from the Phase 2 MaGic trial evaluating the safety and efficacy of claseprubart (DNTH103) in adults with acetylcholine receptor antibody positive (AChR+) generalized Myasthenia Gravis (gMG).
“The results from MaGic mark a significant milestone for Dianthus and are an important step forward for people living with gMG. These results are also a reflection of the commitment and talent of the entire Dianthus team, and I want to thank them for their outstanding execution of this clinical trial,” said Marino Garcia, President and Chief Executive Officer of Dianthus Therapeutics. “The concordance of the efficacy and safety data from the MaGic trial strongly support our best-in-class target product profile for claseprubart 300mg/2mL Q2W in gMG and bolsters our confidence in our execution of the CIDP and MMN clinical programs.”
1

|

|
About the Phase 2 MaGic Trial
The MaGic trial is a global, randomized, double-blind, placebo-controlled Phase 2 trial that enrolled 65 AChR+ participants with gMG. Following an initial loading dose, claseprubart was administered every two weeks (Q2W) via subcutaneous (S.C.) injection at a dose of 300mg/2mL or 600mg/4mL. The initial randomized treatment duration was 13 weeks, followed by a 52-week open-label extension. The primary endpoint of the study was safety and tolerability. Secondary and exploratory efficacy endpoints included Myasthenia Gravis Activities of Daily Living Scale (MG-ADL) and Quantitative Myasthenia Gravis (QMG) score assessments, as well as Minimal Symptom Expression (MSE), Myasthenia Gravis Composite (MGC) score and the Myasthenia Gravis Quality of Life Scale (MG-QOL-15r).
Summary Results from the Phase 2 MaGic Trial
Claseprubart 300mg and 600mg demonstrated rapid, statistically significant and clinically meaningful improvements over placebo as measured by both MG-ADL and QMG, including at Week 1 and at Week 13. The claseprubart 300mg Q2W dose was also statistically significant and clinically meaningful across other key efficacy endpoints, including Minimal Symptom Expression (MSE), Myasthenia Gravis Composite (MGC) Score and the Myasthenia Gravis Quality of Life Scale (MG-QoL-15r).
Claseprubart was generally well tolerated with no drug-related Serious Adverse Events (SAEs) or discontinuations due to any related adverse event. Claseprubart had a favorable clinical safety profile comparable to placebo with no treatment-related serious bacterial infections and no clinical symptoms of emergent autoimmune disorders observed.
“I would like to thank the MaGic investigators and site staff who made the seamless execution of this study possible,” said Simrat Randhawa, MD, Chief Medical Officer of Dianthus Therapeutics. “The consistent and meaningful treatment effect seen in both treatment arms across multiple standard MG efficacy metrics in this Phase 2 trial gives me and my team great confidence in our ability to execute a successful Phase 3 trial.”
“gMG is a chronic condition that can be treated with complement and FcRn inhibitors. The ideal drug should provide continuous symptom control and have low infection risks, infrequent side effects, convenient dosing schedule, and minimal administration burden,” said Dr. Tuan Vu, a Professor of Neurology at the University of South Florida Morsani College of Medicine. “If these impressive results were replicated in a Phase 3 trial, claseprubart may be a differentiated treatment option for patients with gMG.”
Efficacy Results
MG-ADL
| • | Claseprubart dosed at 300mg S.C. Q2W achieved a statistically significant and clinically meaningful mean improvement from baseline of 4.6 points in MG-ADL score at Week 13 (placebo-adjusted improvement: 1.8 points; P=0.0113). A statistically significant improvement in the MG-ADL was also seen as early as Week 1 with 300mg. |
| • | Claseprubart dosed at 600mg S.C. Q2W achieved a statistically significant and clinically meaningful mean improvement from baseline of 5.4 points in MG-ADL score at Week 13 (placebo-adjusted improvement: 2.6 points; P=0.0006). A statistically significant improvement in the MG-ADL was also seen as early as Week 1 with 600mg. |
| • | As expected, there was no statistically significant difference between the claseprubart 300mg and 600mg arms in MG-ADL score at any time point. |
2

|

|
QMG
| • | Claseprubart dosed at 300mg S.C. Q2W achieved a statistically significant and clinically meaningful mean improvement from baseline of 4.4 points in QMG score at Week 13 (placebo-adjusted improvement: 2.4; P=0.0144). A statistically significant improvement in QMG was also seen as early as Week 1 with 300mg. |
| • | Claseprubart dosed at 600mg S.C. Q2W achieved a statistically significant and clinically meaningful mean improvement from baseline of 4.5 points in QMG score at Week 13 (placebo-adjusted improvement: 2.5; P=0.0111). A statistically significant improvement in QMG was also seen as early as Week 1 with 600mg. |
| • | As expected, there was no statistically significant difference between the claseprubart 300mg and 600mg arms in QMG score at any time point. |
Efficacy Summary at Week 13
| Placebo N=22 |
Claseprubart 300mg Q2W N=21 |
Claseprubart 600mg Q2W N=22 |
||||||||||||||||||
| Absolute | Placebo- Adjusted |
Absolute | Placebo- Adjusted |
|||||||||||||||||
| MG-ADL mean change from baseline |
-2.8 | -4.6 | |
-1.8 (P=0.0113)* |
|
-5.4 | |
-2.6 (P=0.0006)* |
| |||||||||||
| QMG mean change from baseline |
-2.0 | -4.4 | |
-2.4 (P=0.0144)* |
|
-4.5 | |
-2.5 (P=0.0111)* |
| |||||||||||
| MSE |
14 | % | 37 | % | |
23% (P=0.0550)* |
|
27 | % | |
13% (P=0.1031) |
| ||||||||
| MGC mean change from baseline |
-3.1 | -8.7 | |
-5.6 (P=0.0008)* |
|
-8.6 | |
-5.5 (P=0.0008)* |
| |||||||||||
| MG-QoL-15r mean change from baseline |
-3.9 | -6.1 | |
-2.2 (P=0.0414)* |
|
-5.4 | |
-1.5 (P=0.1122) |
| |||||||||||
| * | One-sided p-values are presented for comparisons of claseprubart vs placebo, with any p-value below 0.1 considered nominally statistically significant. |
Safety and Tolerability Results
| • | Claseprubart was generally well tolerated and demonstrated a clinical safety profile in both treatment arms comparable to placebo. |
| • | No infection signal was observed, including no related serious bacterial infections in the treatment arms; the only related serious adverse event (SAE) of infection occurred in the placebo arm. |
| • | There were no symptoms indicative of autoimmune activation. |
| • | Injection site reactions (ISRs) were infrequent and generally mild, and there were no claseprubart discontinuations from RCT due to related AEs. |
3

|

|
Next Steps
“Based on the comparable efficacy data demonstrated in MaGic by the 300mg and 600mg Q2W doses, as well as encouraging early data from the open-label extension, we are planning for an end-of-Phase 2 meeting with the FDA to align on the proposed design of a Phase 3 trial for claseprubart in Myasthenia Gravis that investigates both 300mg Q2W and 300mg Q4W doses vs. placebo,” said Marino Garcia, President and Chief Executive Officer of Dianthus Therapeutics. “2026 will be a catalyst-rich year, with the initiation of the Phase 3 gMG trial followed by results from the interim responder analysis of the Phase 3 CAPTIVATE trial in Chronic Inflammatory Demyelinating Polyneuropathy (CIDP) and top-line results from the Phase 2 MoMeNtum trial in Multifocal Motor Neuropathy (MMN), both in 2H’26.”
Investor Conference Call & Webcast to be Held at 8:00 a.m. ET Today
Dianthus Therapeutics will host an investor call and webcast to discuss the Phase 2 MaGic results today, September 8, 2025 at 8:00 a.m. ET. Please click here to register for this event.
The live webcast may be accessed via the Investors section of the Dianthus Therapeutics website at https://investor.dianthustx.com/. A replay of the webcast will be available following the call.
About Claseprubart (DNTH103)
Claseprubart is an investigational, clinical-stage, potent monoclonal antibody engineered to selectively target the classical pathway by inhibiting only the active form of the C1s protein, a clinically validated complement target. Claseprubart is enhanced with YTE half-life extension technology designed to enable a more convenient subcutaneous, infrequently dosed, self-administered injection. Additionally, selective inhibition of the classical complement pathway may lower patient risk of infection from encapsulated bacteria by preserving immune activity of the lectin and alternative pathways. As the classical pathway plays a significant role in disease pathology, claseprubart has the potential to be a best-in-class pipeline-in-a-product across a range of autoimmune disorders with high unmet need. Dianthus is building a neuromuscular franchise with claseprubart and expects to initiate a Phase 3 trial in gMG in 2026, the interim responder analysis of the Phase 3 CAPTIVATE trial in Chronic Inflammatory Demyelinating Polyneuropathy in 2H’26, and top-line data from the Phase 2 MoMeNtum trial in Multifocal Motor Neuropathy in 2H’26.
Claseprubart is an investigational agent that is not approved as a therapy in any indication in any jurisdiction worldwide.
About Generalized Myasthenia Gravis
Generalized Myasthenia Gravis (gMG) is a chronic autoimmune disorder driven by the classical pathway that causes progressive muscle weakness. Over 100,000 people in the U.S. are living with gMG and approximately 85% have AChR autoantibody-driven disease. Despite availability of current treatment options, a significant number of patients remain uncontrolled and are seeking better treatment options which may offer sustained efficacy, lower potential risk for infections, and convenient dosing and administration.
About Dianthus Therapeutics
Dianthus Therapeutics is a clinical-stage biotechnology company dedicated to designing and delivering novel, best-in-class monoclonal antibodies with improved selectivity and potency. Based in New York City and Waltham, Mass., Dianthus is comprised of an experienced team of biotech and pharma executives who are leading the development of next-generation antibody complement therapeutics, aiming to deliver transformative medicines for people living with severe autoimmune and inflammatory diseases.
4

|

|
To learn more, please visit www.dianthustx.com and follow us on LinkedIn.
Cautionary Statement Regarding Forward-Looking Statements
Certain statements in this press release, other than purely historical information, may constitute “forward-looking statements” within the meaning of the federal securities laws, including for purposes of the safe harbor provisions under the United States Private Securities Litigation Reform Act of 1995, express or implied statements regarding future plans and prospects, including statements regarding the expectations or plans for discovery, preclinical studies, clinical trials and research and development programs, in particular with respect to claseprubart, and any developments or results in connection therewith, including the target product profile and administration of claseprubart; the anticipated timing of the initiation and results from those studies and trials; expectations regarding the clinical trial design for the Phase 3 trial for claseprubart in gMG; expectations regarding the time period over which the Company’s capital resources are expected to be sufficient to fund its anticipated operations; and expectations regarding market size, patient population size, and potential opportunities for complement therapies, in particular with respect to claseprubart. Claseprubart is an investigational agent that is not approved as a therapy in any indication in any jurisdiction worldwide. The words “opportunity,” “potential,” “milestones,” “runway,” “will,” “anticipate,” “achieve,” “near-term,” “catalysts,” “pursue,” “pipeline,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “might,” “plan,” “possible,” “predict,” “project,” “should,” “strive,” “would,” “aim,” “target,” “commit,” and similar expressions (including the negatives of these terms or variations of them) generally identify forward-looking statements, but the absence of these words does not mean that statement is not forward looking.
Actual results could differ materially from those included in the forward-looking statements due to various factors, risks and uncertainties, including, but not limited to, that preclinical testing of claseprubart and data from clinical trials may not be predictive of the results or success of ongoing or later clinical trials, that the development of claseprubart or the Company’s other compounds may take longer and/or cost more than planned, that the Company may be unable to successfully complete the clinical development of the Company’s compounds, that the Company may be delayed in initiating, enrolling or completing its planned clinical trials, and that the Company’s compounds may not receive regulatory approval or become commercially successful products. These and other risks and uncertainties are identified under the heading “Risk Factors” included in the Company’s Annual Report on Form 10-K for the period ended December 31, 2024, and other filings that the Company has made and may make with the SEC in the future. Nothing in this press release should be regarded as a representation by any person that the forward-looking statements set forth herein will be achieved or that any of the contemplated results of such forward-looking statements will be achieved.
The forward-looking statements in this press release speak only as of the date they are made and are qualified in their entirety by reference to the cautionary statements herein. Dianthus undertakes no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events or otherwise, except as required by law.
Contact
Jennifer Davis Ruff
Dianthus Therapeutics
jdavisruff@dianthustx.com
5
Claseprubart (DNTH103) Top-line Ph. 2 MaGic Results in Generalized Myasthenia Gravis September 8, 2025 Exhibit 99.2

Forward-looking statements Certain statements in this presentation (“Presentation”), other than purely historical information, may constitute “forward-looking statements” within the meaning of the federal securities laws, including for purposes of the safe harbor provisions under the United States Private Securities Litigation Reform Act of 1995, concerning Dianthus Therapeutics, Inc. (the “Company”). These forward-looking statements include statements regarding the Company’s future plans and prospects, including statements regarding the expectations or plans for discovery, preclinical studies, clinical trials and research and development programs, in particular with respect to claseprubart, and any developments or results in connection therewith, including the target product profile and administration of claseprubart; the anticipated timing of the results from those studies and trials; expectations regarding the clinical trial design for the Phase 3 trial for claseprubart; expectations regarding the use of proceeds and the time period over which the Company’s capital resources will be sufficient to fund its anticipated operations; and expectations regarding market size, patient population size and potential opportunities for complement therapies, in particular with respect to claseprubart. Claseprubart is an investigational agent that is not approved as a therapy in any indication in any jurisdiction worldwide. The words “opportunity,” “potential,” “milestones,” “runway,” “will,” “anticipate,” “achieve,” “near-term,” “catalysts,” “pursue,” “pipeline,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “might,” “plan,” “possible,” “predict,” “project,” “should,” “strive,” “would,” “aim,” “target,” “commit,” and similar expressions (including the negatives of these terms or variations of them) generally identify forward-looking statements, but the absence of these words does not mean that statement is not forward looking. Actual results could differ materially from those included in the forward-looking statements due to various factors, risks and uncertainties, including, but not limited to, that preclinical testing of claseprubart and data from clinical trials may not be predictive of the results or success of ongoing or later clinical trials, that the development of claseprubart or the Company's compounds may take longer and/or cost more than planned, that the Company may be unable to successfully complete the clinical development of the Company’s compounds, that the Company may be delayed in initiating, enrolling or completing any clinical trials, and that the Company's compounds may not receive regulatory approval or become commercially successful products. These and other risks and uncertainties are identified under the heading "Risk Factors" included in the Company’s Annual Report on Form 10-K for the period ended December 31, 2024, and other filings that the Company has made and may make with the SEC in the future. Nothing in this Presentation should be regarded as a representation by any person that the forward-looking statements set forth herein will be achieved or that any of the contemplated results of such forward-looking statements will be achieved. Dianthus undertakes no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events or otherwise, except as required by law.

Introduction Marino Garcia, Chief Executive Officer MaGic Phase 2 Top-line Results Simrat Randhawa, MD, Chief Medical Officer Claseprubart Summary Remarks Marino Garcia, Chief Executive Officer Analyst Q&A Marino Garcia, Chief Executive Officer Simrat Randhawa, MD, Chief Medical Officer John King, Chief Commercial Officer Ryan Savitz, Chief Financial Officer & Chief Business Officer Agenda

Introduction Marino Garcia, CEO
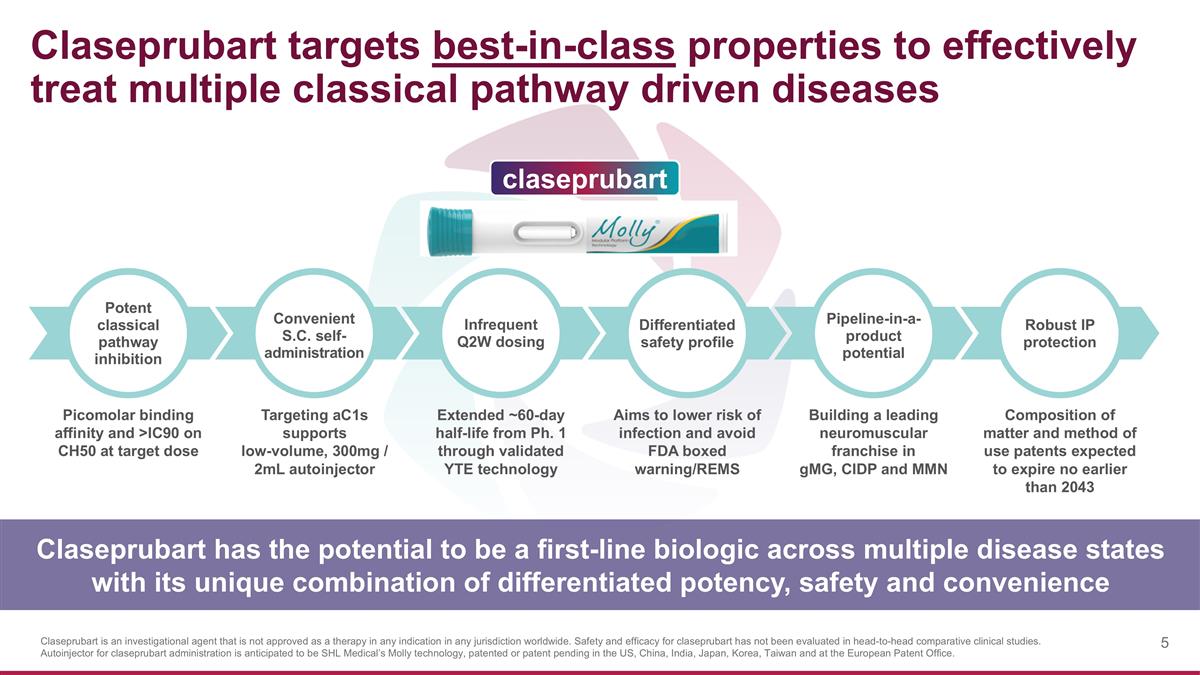
Claseprubart targets best-in-class properties to effectively treat multiple classical pathway driven diseases Picomolar binding affinity and >IC90 on CH50 at target dose Extended ~60-day half-life from Ph. 1 through validated YTE technology Targeting aC1s supports low-volume, 300mg / 2mL autoinjector Aims to lower risk of infection and avoid FDA boxed warning/REMS Building a leading neuromuscular franchise in gMG, CIDP and MMN Composition of matter and method of use patents expected to expire no earlier than 2043 Infrequent Q2W dosing Convenient S.C. self- administration Potent classical pathway inhibition Differentiated safety profile Pipeline-in-a-product potential Robust IP protection Claseprubart has the potential to be a first-line biologic across multiple disease states with its unique combination of differentiated potency, safety and convenience claseprubart Claseprubart is an investigational agent that is not approved as a therapy in any indication in any jurisdiction worldwide. Safety and efficacy for claseprubart has not been evaluated in head-to-head comparative clinical studies. Autoinjector for claseprubart administration is anticipated to be SHL Medical’s Molly technology, patented or patent pending in the US, China, India, Japan, Korea, Taiwan and at the European Patent Office.
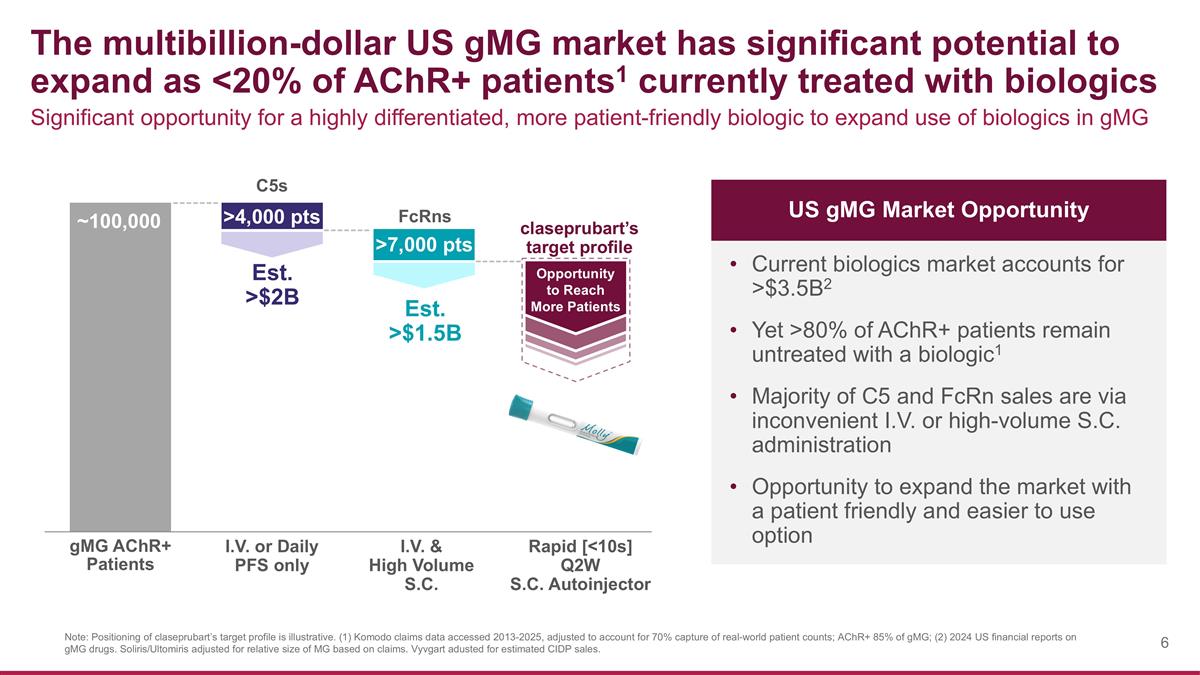
The multibillion-dollar US gMG market has significant potential to expand as <20% of AChR+ patients1 currently treated with biologics Note: Positioning of claseprubart’s target profile is illustrative. (1) Komodo claims data accessed 2013-2025, adjusted to account for 70% capture of real-world patient counts; AChR+ 85% of gMG; (2) 2024 US financial reports on gMG drugs. Soliris/Ultomiris adjusted for relative size of MG based on claims. Vyvgart adusted for estimated CIDP sales. ~100,000 Significant opportunity for a highly differentiated, more patient-friendly biologic to expand use of biologics in gMG Current biologics market accounts for >$3.5B2 Yet >80% of AChR+ patients remain untreated with a biologic1 Majority of C5 and FcRn sales are via inconvenient I.V. or high-volume S.C. administration Opportunity to expand the market with a patient friendly and easier to use option I.V. or Daily PFS only I.V. & High Volume S.C. Rapid [<10s] Q2W S.C. Autoinjector C5s FcRns claseprubart’s target profile Est. >$2B Est. >$1.5B Opportunity to Reach More Patients >4,000 pts >7,000 pts US gMG Market Opportunity
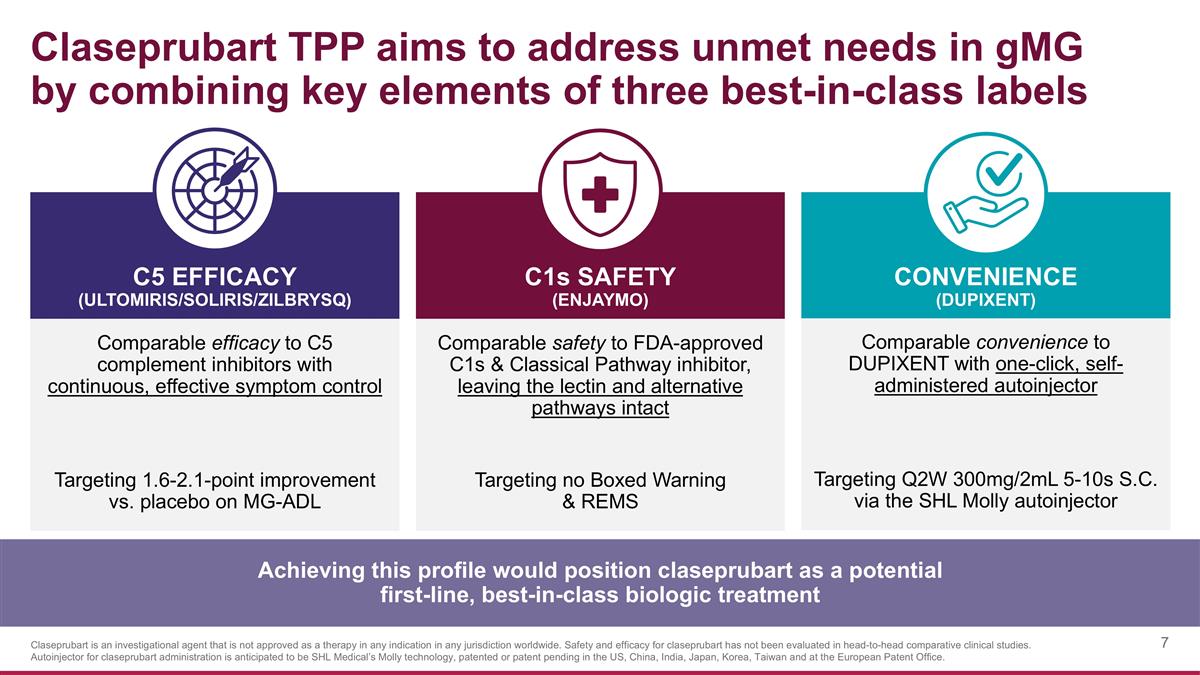
Claseprubart TPP aims to address unmet needs in gMG by combining key elements of three best-in-class labels Comparable efficacy to C5 complement inhibitors with continuous, effective symptom control Targeting 1.6-2.1-point improvement vs. placebo on MG-ADL C1s SAFETY (Enjaymo) C5 EFFICACY (ULTOMIRIS/SOLIRIS/ZILBRYSQ) CONVENIENCE (DUPIXENT) Comparable safety to FDA-approved C1s & Classical Pathway inhibitor, leaving the lectin and alternative pathways intact Targeting no Boxed Warning & REMS Comparable convenience to DUPIXENT with one-click, self-administered autoinjector Targeting Q2W 300mg/2mL 5-10s S.C. via the SHL Molly autoinjector Achieving this profile would position claseprubart as a potential first-line, best-in-class biologic treatment Claseprubart is an investigational agent that is not approved as a therapy in any indication in any jurisdiction worldwide. Safety and efficacy for claseprubart has not been evaluated in head-to-head comparative clinical studies. Autoinjector for claseprubart administration is anticipated to be SHL Medical’s Molly technology, patented or patent pending in the US, China, India, Japan, Korea, Taiwan and at the European Patent Office.
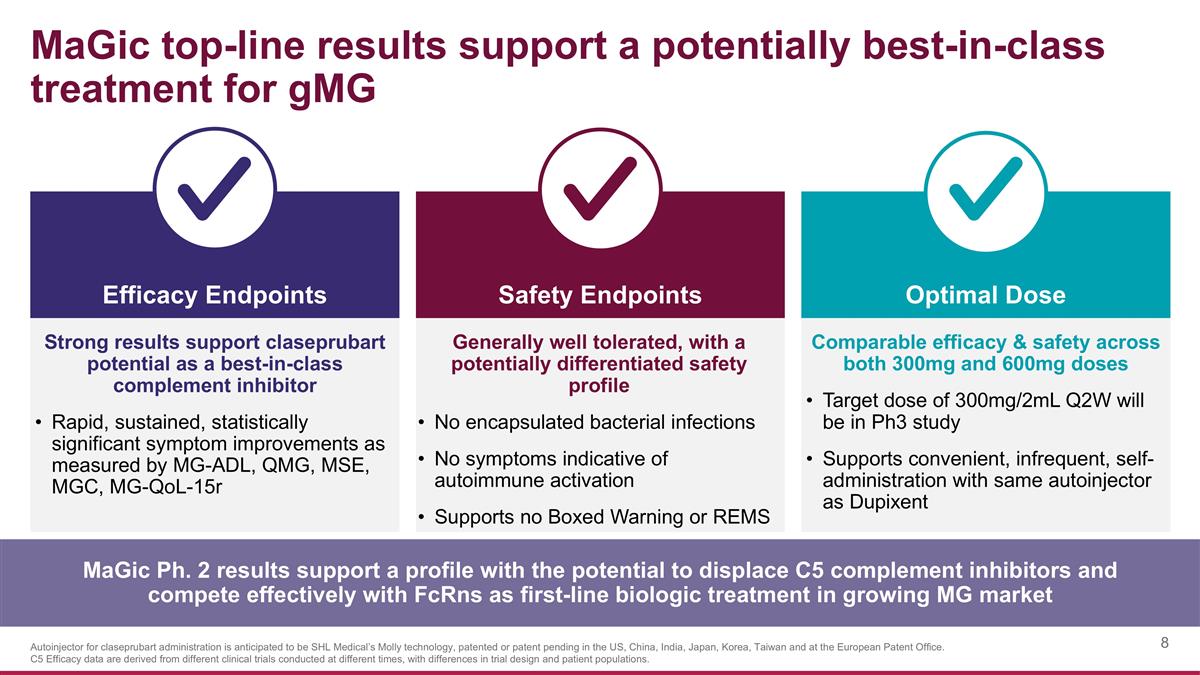
MaGic top-line results support a potentially best-in-class treatment for gMG Strong results support claseprubart potential as a best-in-class complement inhibitor Rapid, sustained, statistically significant symptom improvements as measured by MG-ADL, QMG, MSE, MGC, MG-QoL-15r Safety Endpoints Efficacy Endpoints Optimal Dose Generally well tolerated, with a potentially differentiated safety profile No encapsulated bacterial infections No symptoms indicative of autoimmune activation Supports no Boxed Warning or REMS Comparable efficacy & safety across both 300mg and 600mg doses Target dose of 300mg/2mL Q2W will be in Ph3 study Supports convenient, infrequent, self-administration with same autoinjector as Dupixent MaGic Ph. 2 results support a profile with the potential to displace C5 complement inhibitors and compete effectively with FcRns as first-line biologic treatment in growing MG market Autoinjector for claseprubart administration is anticipated to be SHL Medical’s Molly technology, patented or patent pending in the US, China, India, Japan, Korea, Taiwan and at the European Patent Office. C5 Efficacy data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations.

MaGic Phase 2 Top-line Results Simrat Randhawa, MD, CMO
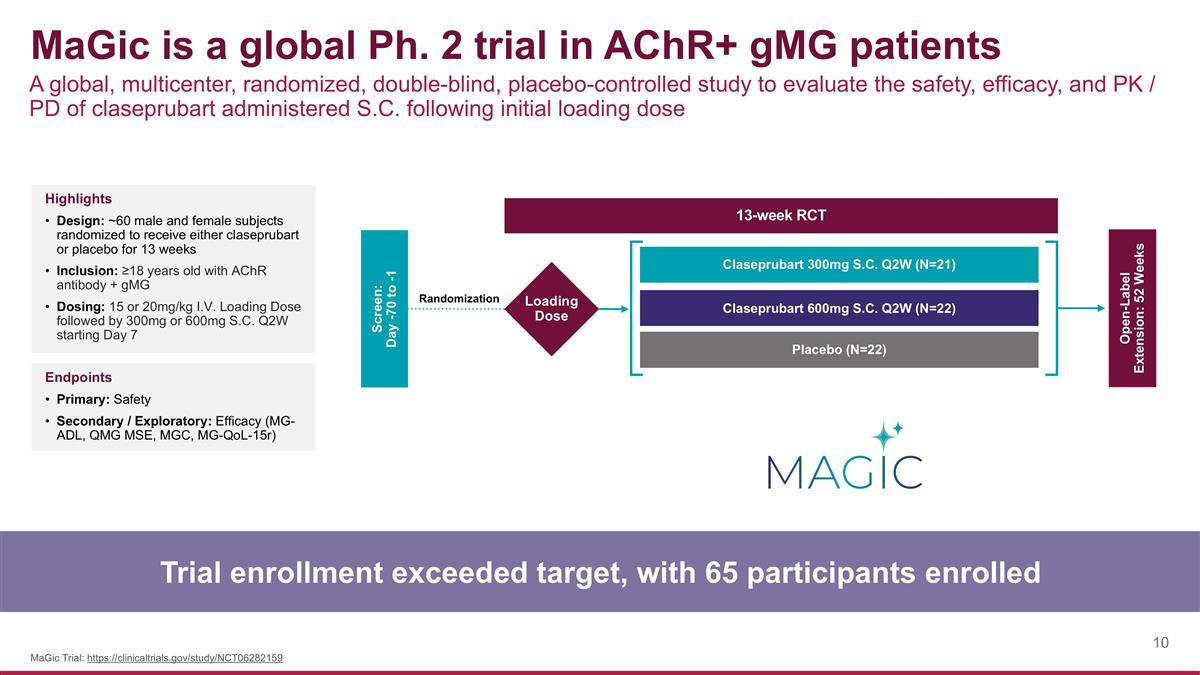
MaGic is a global Ph. 2 trial in AChR+ gMG patients A global, multicenter, randomized, double-blind, placebo-controlled study to evaluate the safety, efficacy, and PK / PD of claseprubart administered S.C. following initial loading dose Trial enrollment exceeded target, with 65 participants enrolled MaGic Trial: https://clinicaltrials.gov/study/NCT06282159 Highlights Design: ~60 male and female subjects randomized to receive either claseprubart or placebo for 13 weeks Inclusion: ≥18 years old with AChR antibody + gMG Dosing: 15 or 20mg/kg I.V. Loading Dose followed by 300mg or 600mg S.C. Q2W starting Day 7 Endpoints Primary: Safety Secondary / Exploratory: Efficacy (MG-ADL, QMG MSE, MGC, MG-QoL-15r) Screen: Day -70 to -1 Open-Label Extension: 52 Weeks Claseprubart 300mg S.C. Q2W (N=21) Claseprubart 600mg S.C. Q2W (N=22) Placebo (N=22) 13-week RCT Loading Dose Randomization
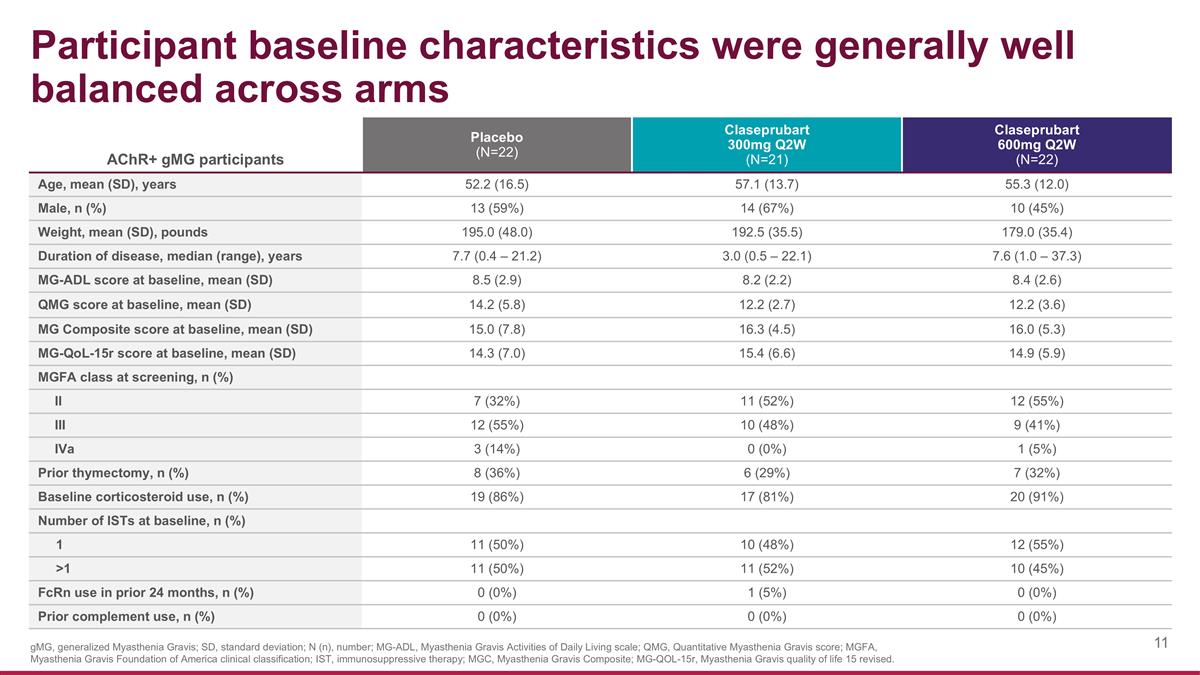
AChR+ gMG participants Placebo (N=22) Claseprubart 300mg Q2W (N=21) Claseprubart 600mg Q2W (N=22) Age, mean (SD), years 52.2 (16.5) 57.1 (13.7) 55.3 (12.0) Male, n (%) 13 (59%) 14 (67%) 10 (45%) Weight, mean (SD), pounds 195.0 (48.0) 192.5 (35.5) 179.0 (35.4) Duration of disease, median (range), years 7.7 (0.4 – 21.2) 3.0 (0.5 – 22.1) 7.6 (1.0 – 37.3) MG-ADL score at baseline, mean (SD) 8.5 (2.9) 8.2 (2.2) 8.4 (2.6) QMG score at baseline, mean (SD) 14.2 (5.8) 12.2 (2.7) 12.2 (3.6) MG Composite score at baseline, mean (SD) 15.0 (7.8) 16.3 (4.5) 16.0 (5.3) MG-QoL-15r score at baseline, mean (SD) 14.3 (7.0) 15.4 (6.6) 14.9 (5.9) MGFA class at screening, n (%) II 7 (32%) 11 (52%) 12 (55%) III 12 (55%) 10 (48%) 9 (41%) IVa 3 (14%) 0 (0%) 1 (5%) Prior thymectomy, n (%) 8 (36%) 6 (29%) 7 (32%) Baseline corticosteroid use, n (%) 19 (86%) 17 (81%) 20 (91%) Number of ISTs at baseline, n (%) 1 11 (50%) 10 (48%) 12 (55%) >1 11 (50%) 11 (52%) 10 (45%) FcRn use in prior 24 months, n (%) 0 (0%) 1 (5%) 0 (0%) Prior complement use, n (%) 0 (0%) 0 (0%) 0 (0%) Participant baseline characteristics were generally well balanced across arms gMG, generalized Myasthenia Gravis; SD, standard deviation; N (n), number; MG-ADL, Myasthenia Gravis Activities of Daily Living scale; QMG, Quantitative Myasthenia Gravis score; MGFA, Myasthenia Gravis Foundation of America clinical classification; IST, immunosuppressive therapy; MGC, Myasthenia Gravis Composite; MG-QOL-15r, Myasthenia Gravis quality of life 15 revised.
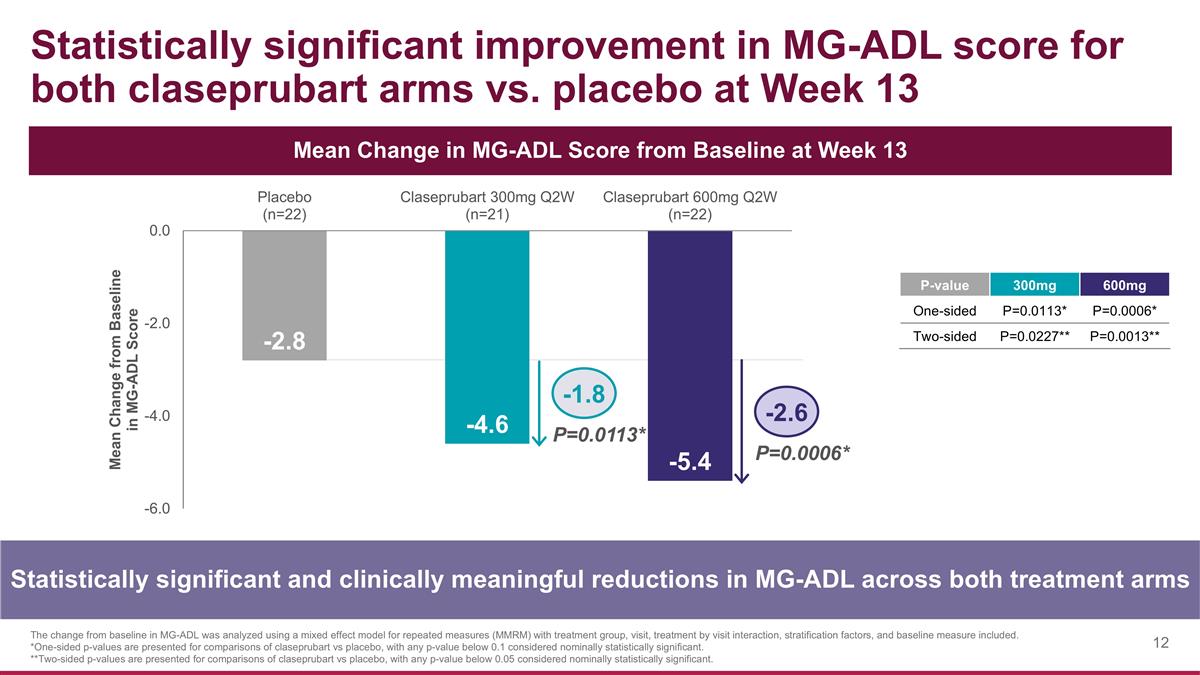
Statistically significant improvement in MG-ADL score for both claseprubart arms vs. placebo at Week 13 Statistically significant and clinically meaningful reductions in MG-ADL across both treatment arms Mean Change in MG-ADL Score from Baseline at Week 13 The change from baseline in MG-ADL was analyzed using a mixed effect model for repeated measures (MMRM) with treatment group, visit, treatment by visit interaction, stratification factors, and baseline measure included. *One-sided p-values are presented for comparisons of claseprubart vs placebo, with any p-value below 0.1 considered nominally statistically significant. **Two-sided p-values are presented for comparisons of claseprubart vs placebo, with any p-value below 0.05 considered nominally statistically significant. P=0.0113* P=0.0006* -1.8 -2.6 P-value 300mg 600mg One-sided P=0.0113* P=0.0006* Two-sided P=0.0227** P=0.0013**
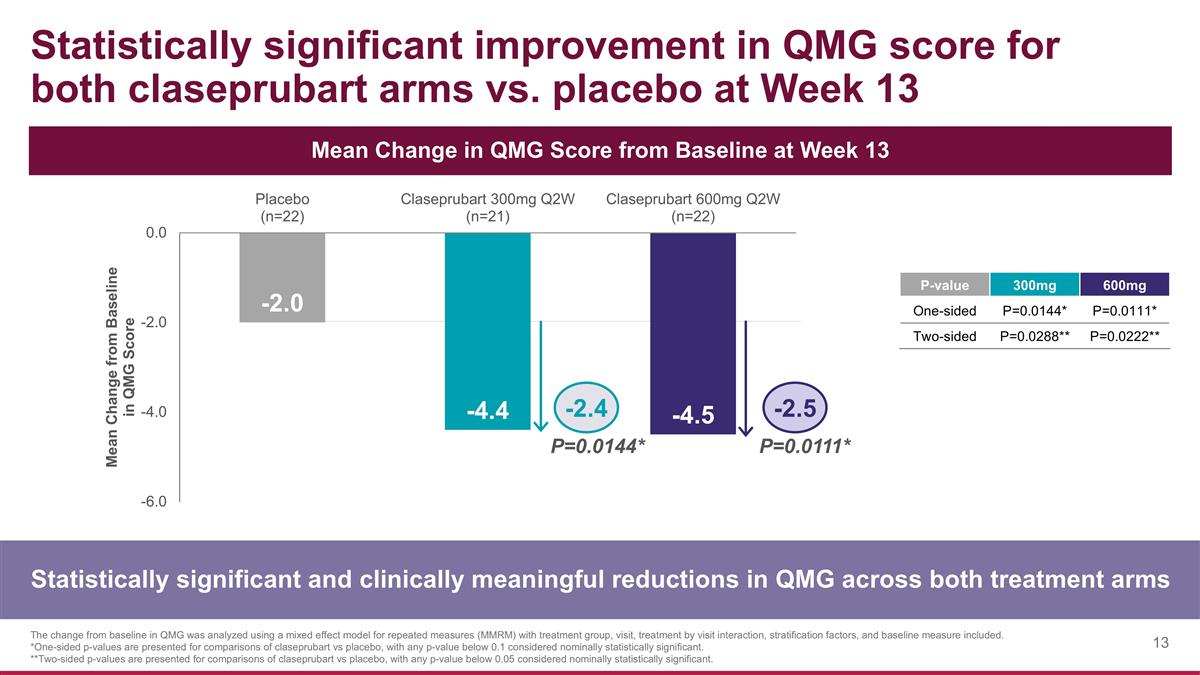
Statistically significant improvement in QMG score for both claseprubart arms vs. placebo at Week 13 Statistically significant and clinically meaningful reductions in QMG across both treatment arms The change from baseline in QMG was analyzed using a mixed effect model for repeated measures (MMRM) with treatment group, visit, treatment by visit interaction, stratification factors, and baseline measure included. *One-sided p-values are presented for comparisons of claseprubart vs placebo, with any p-value below 0.1 considered nominally statistically significant. **Two-sided p-values are presented for comparisons of claseprubart vs placebo, with any p-value below 0.05 considered nominally statistically significant. Mean Change in QMG Score from Baseline at Week 13 P=0.0144* P=0.0111* -2.4 -2.5 P-value 300mg 600mg One-sided P=0.0144* P=0.0111* Two-sided P=0.0288** P=0.0222**
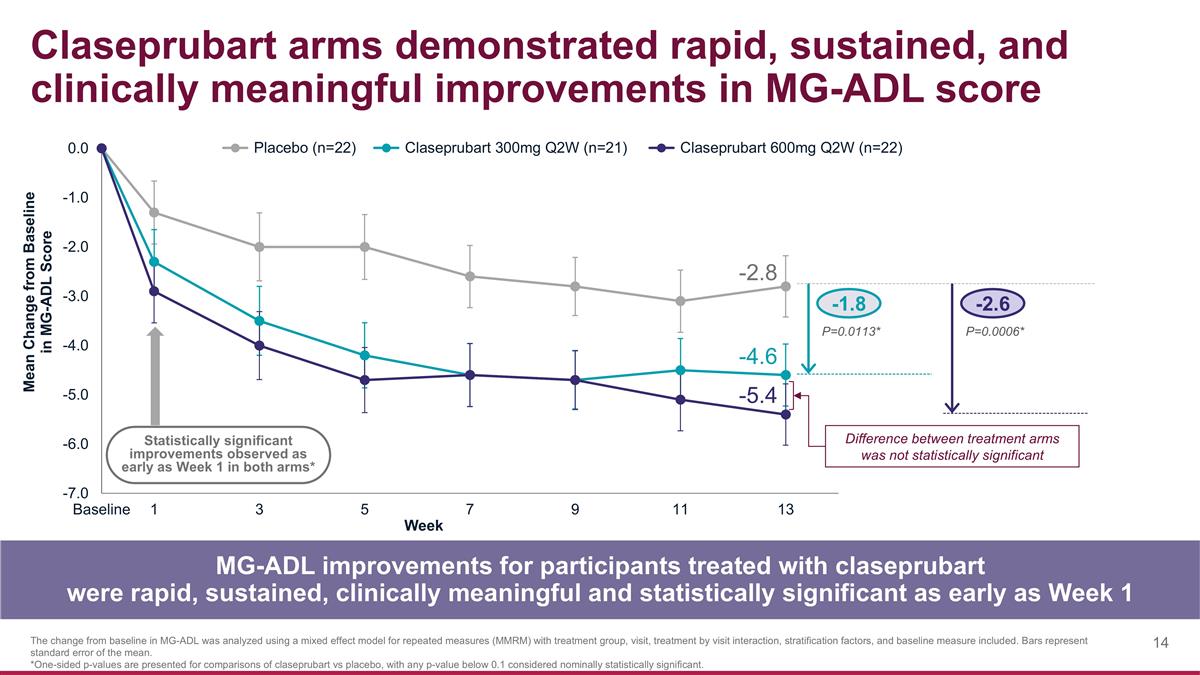
-1.8 -2.6 P=0.0113* P=0.0006* Claseprubart arms demonstrated rapid, sustained, and clinically meaningful improvements in MG-ADL score MG-ADL improvements for participants treated with claseprubart were rapid, sustained, clinically meaningful and statistically significant as early as Week 1 The change from baseline in MG-ADL was analyzed using a mixed effect model for repeated measures (MMRM) with treatment group, visit, treatment by visit interaction, stratification factors, and baseline measure included. Bars represent standard error of the mean. *One-sided p-values are presented for comparisons of claseprubart vs placebo, with any p-value below 0.1 considered nominally statistically significant. Statistically significant improvements observed as early as Week 1 in both arms* -2.8 -4.6 -5.4 Difference between treatment arms was not statistically significant
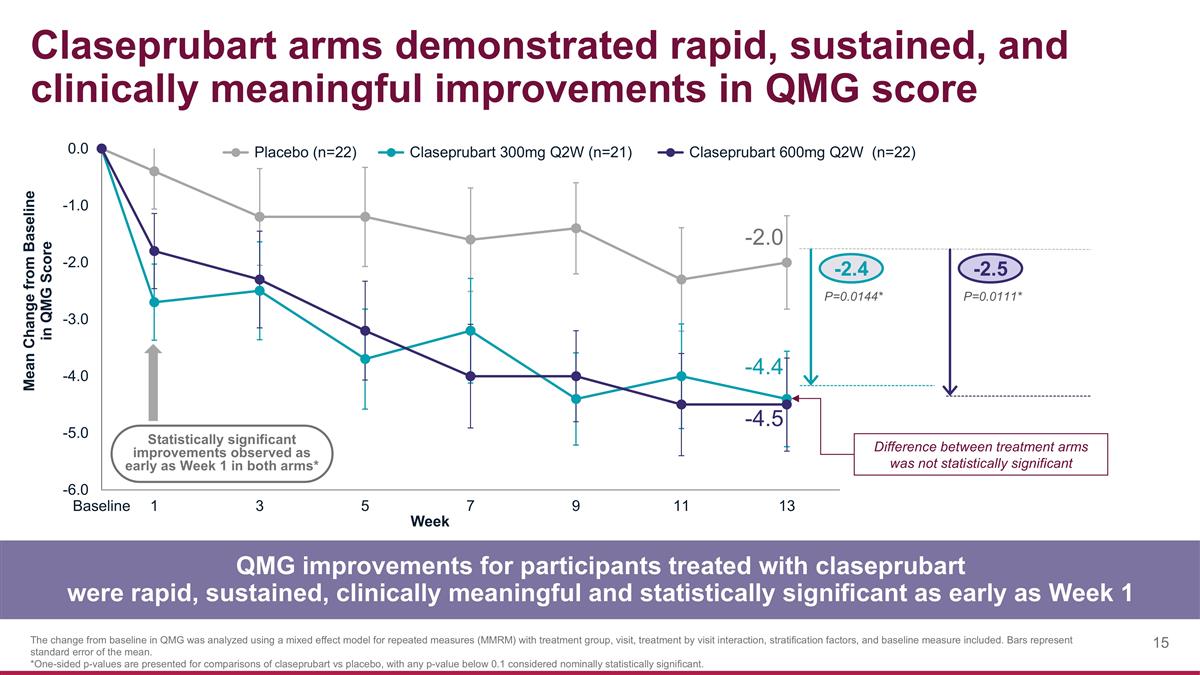
Claseprubart arms demonstrated rapid, sustained, and clinically meaningful improvements in QMG score QMG improvements for participants treated with claseprubart were rapid, sustained, clinically meaningful and statistically significant as early as Week 1 The change from baseline in QMG was analyzed using a mixed effect model for repeated measures (MMRM) with treatment group, visit, treatment by visit interaction, stratification factors, and baseline measure included. Bars represent standard error of the mean. *One-sided p-values are presented for comparisons of claseprubart vs placebo, with any p-value below 0.1 considered nominally statistically significant. -2.4 -2.5 P=0.0144* P=0.0111* -2.0 -4.4 -4.5 Statistically significant improvements observed as early as Week 1 in both arms* Difference between treatment arms was not statistically significant
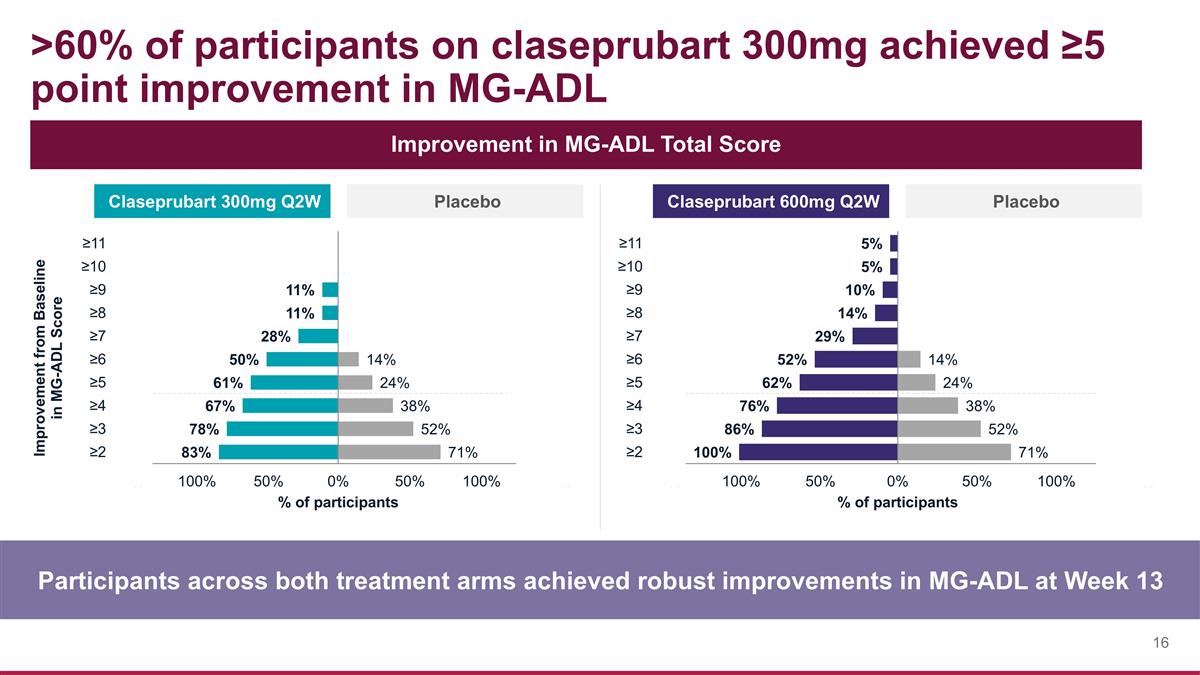
>60% of participants on claseprubart 300mg achieved ≥5 point improvement in MG-ADL Participants across both treatment arms achieved robust improvements in MG-ADL at Week 13 Improvement in MG-ADL Total Score Claseprubart 300mg Q2W Placebo Claseprubart 600mg Q2W Placebo
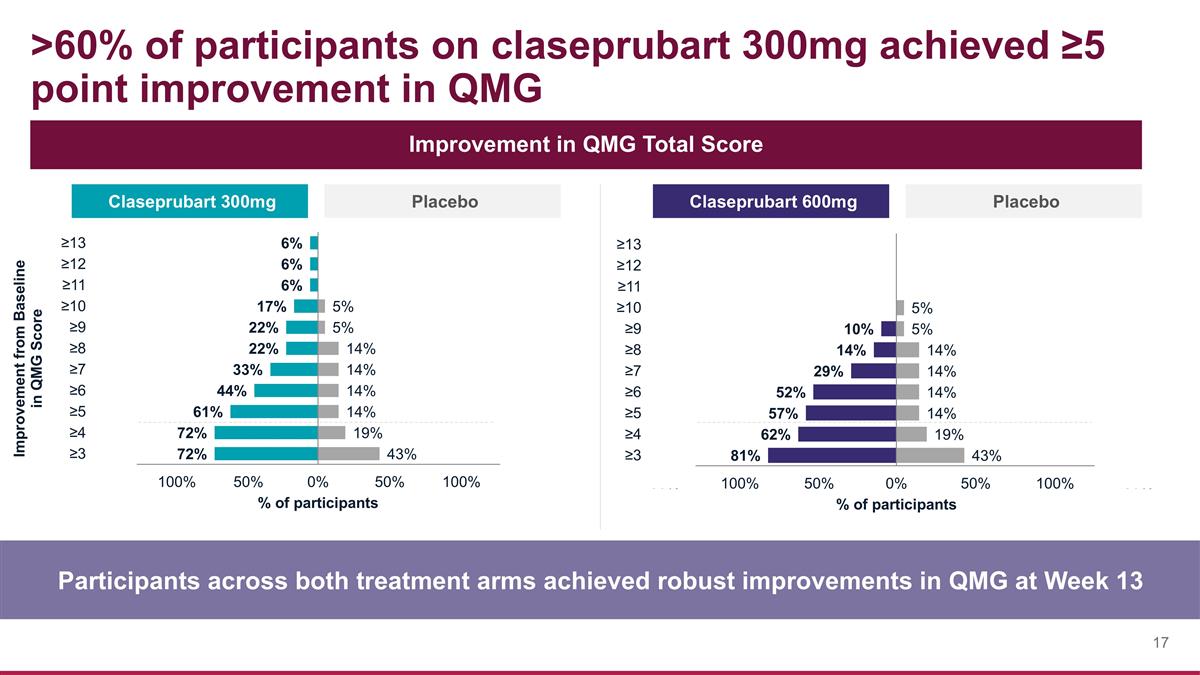
>60% of participants on claseprubart 300mg achieved ≥5 point improvement in QMG Participants across both treatment arms achieved robust improvements in QMG at Week 13 Improvement in QMG Total Score Claseprubart 300mg Placebo Claseprubart 600mg Placebo
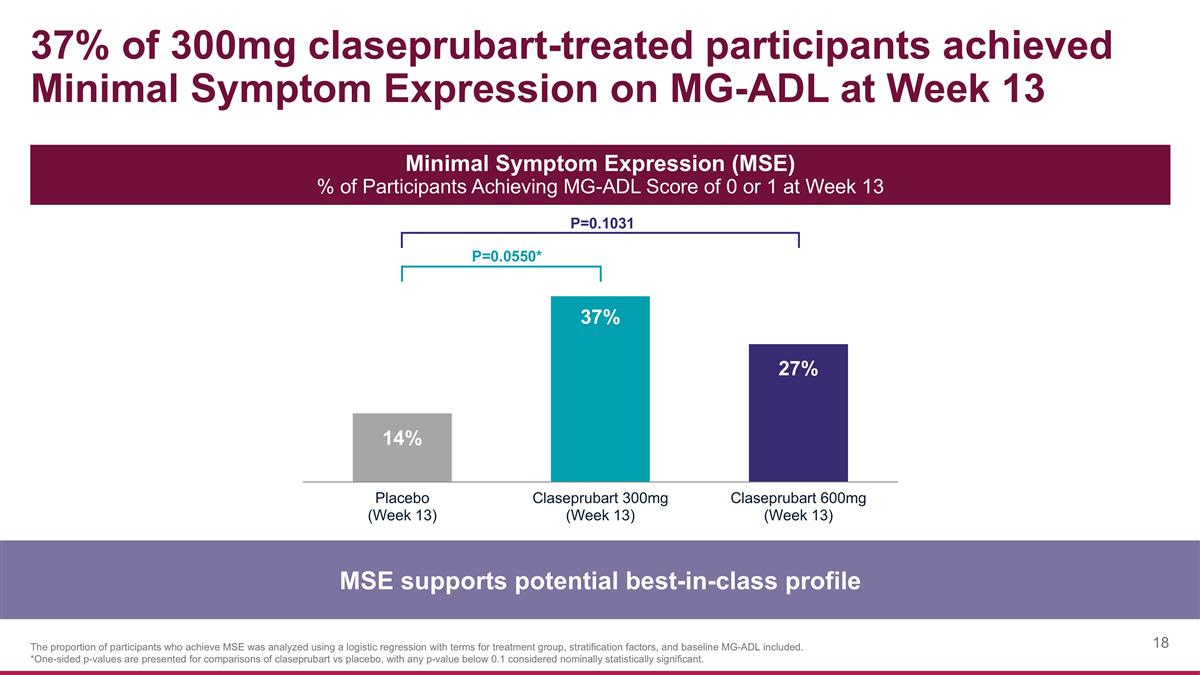
37% of 300mg claseprubart-treated participants achieved Minimal Symptom Expression on MG-ADL at Week 13 MSE supports potential best-in-class profile Minimal Symptom Expression (MSE) % of Participants Achieving MG-ADL Score of 0 or 1 at Week 13 P=0.0550* P=0.1031 The proportion of participants who achieve MSE was analyzed using a logistic regression with terms for treatment group, stratification factors, and baseline MG-ADL included. *One-sided p-values are presented for comparisons of claseprubart vs placebo, with any p-value below 0.1 considered nominally statistically significant.
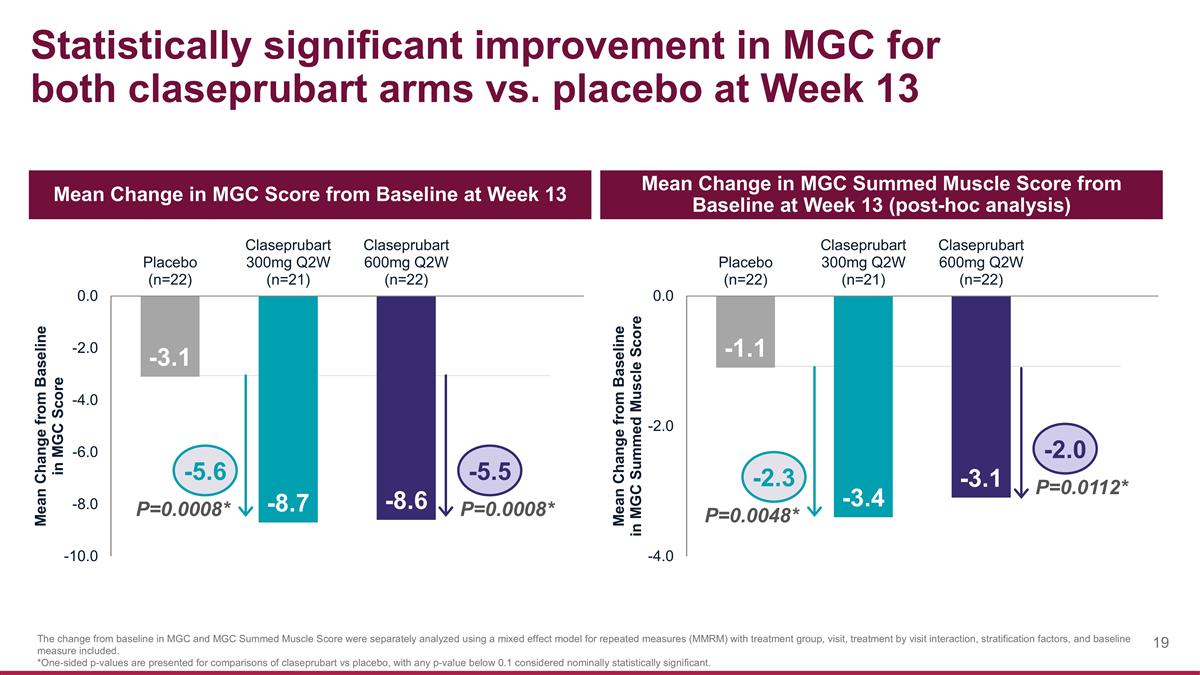
Statistically significant improvement in MGC for both claseprubart arms vs. placebo at Week 13 Mean Change in MGC Score from Baseline at Week 13 Mean Change in MGC Summed Muscle Score from Baseline at Week 13 (post-hoc analysis) The change from baseline in MGC and MGC Summed Muscle Score were separately analyzed using a mixed effect model for repeated measures (MMRM) with treatment group, visit, treatment by visit interaction, stratification factors, and baseline measure included. *One-sided p-values are presented for comparisons of claseprubart vs placebo, with any p-value below 0.1 considered nominally statistically significant. P=0.0008* -5.5 -5.6 P=0.0008* P=0.0112* -2.0 -2.3 P=0.0048*
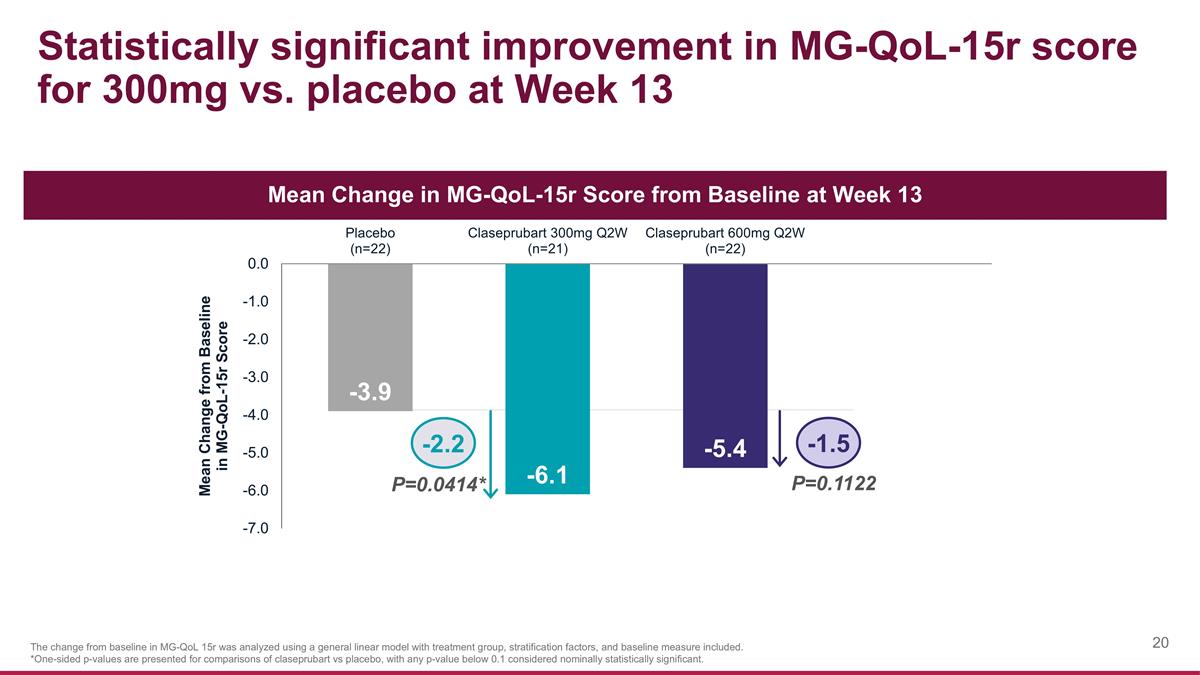
Statistically significant improvement in MG-QoL-15r score for 300mg vs. placebo at Week 13 The change from baseline in MG-QoL 15r was analyzed using a general linear model with treatment group, stratification factors, and baseline measure included. *One-sided p-values are presented for comparisons of claseprubart vs placebo, with any p-value below 0.1 considered nominally statistically significant. P=0.0414* P=0.1122 -2.2 -1.5 Mean Change in MG-QoL-15r Score from Baseline at Week 13
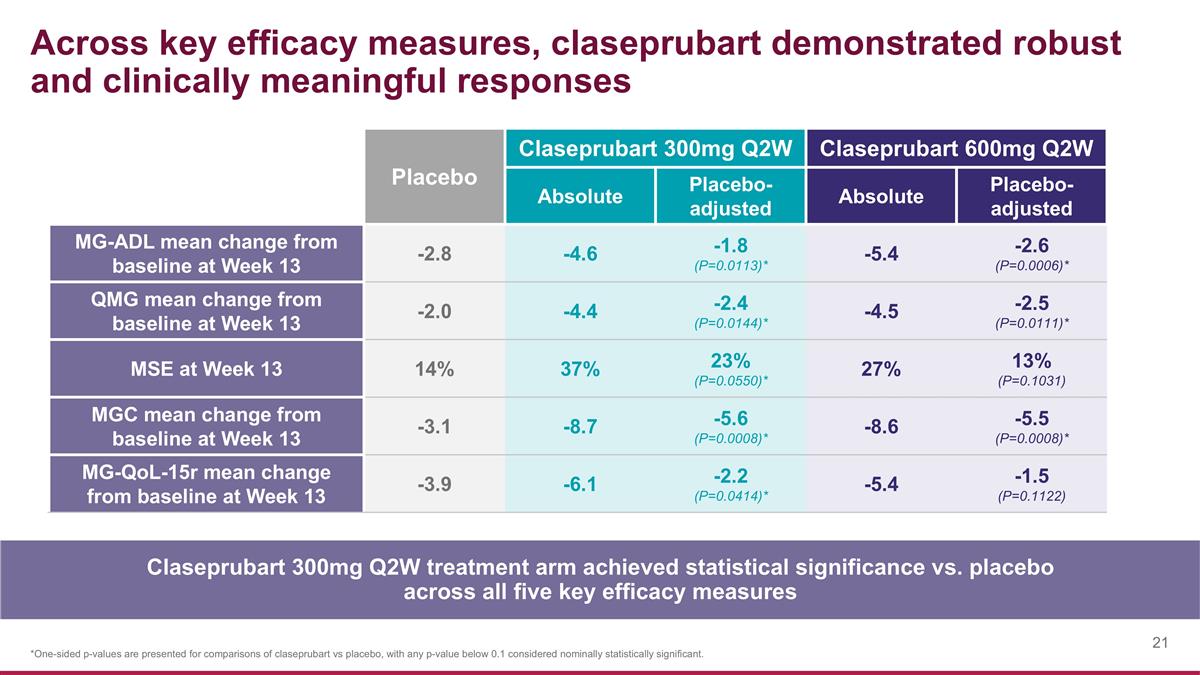
Across key efficacy measures, claseprubart demonstrated robust and clinically meaningful responses Placebo Claseprubart 300mg Q2W Claseprubart 600mg Q2W Absolute Placebo-adjusted Absolute Placebo-adjusted MG-ADL mean change from baseline at Week 13 -2.8 -4.6 -1.8 (P=0.0113)* -5.4 -2.6 (P=0.0006)* QMG mean change from baseline at Week 13 -2.0 -4.4 -2.4 (P=0.0144)* -4.5 -2.5 (P=0.0111)* MSE at Week 13 14% 37% 23% (P=0.0550)* 27% 13% (P=0.1031) MGC mean change from baseline at Week 13 -3.1 -8.7 -5.6 (P=0.0008)* -8.6 -5.5 (P=0.0008)* MG-QoL-15r mean change from baseline at Week 13 -3.9 -6.1 -2.2 (P=0.0414)* -5.4 -1.5 (P=0.1122) *One-sided p-values are presented for comparisons of claseprubart vs placebo, with any p-value below 0.1 considered nominally statistically significant. Claseprubart 300mg Q2W treatment arm achieved statistical significance vs. placebo across all five key efficacy measures
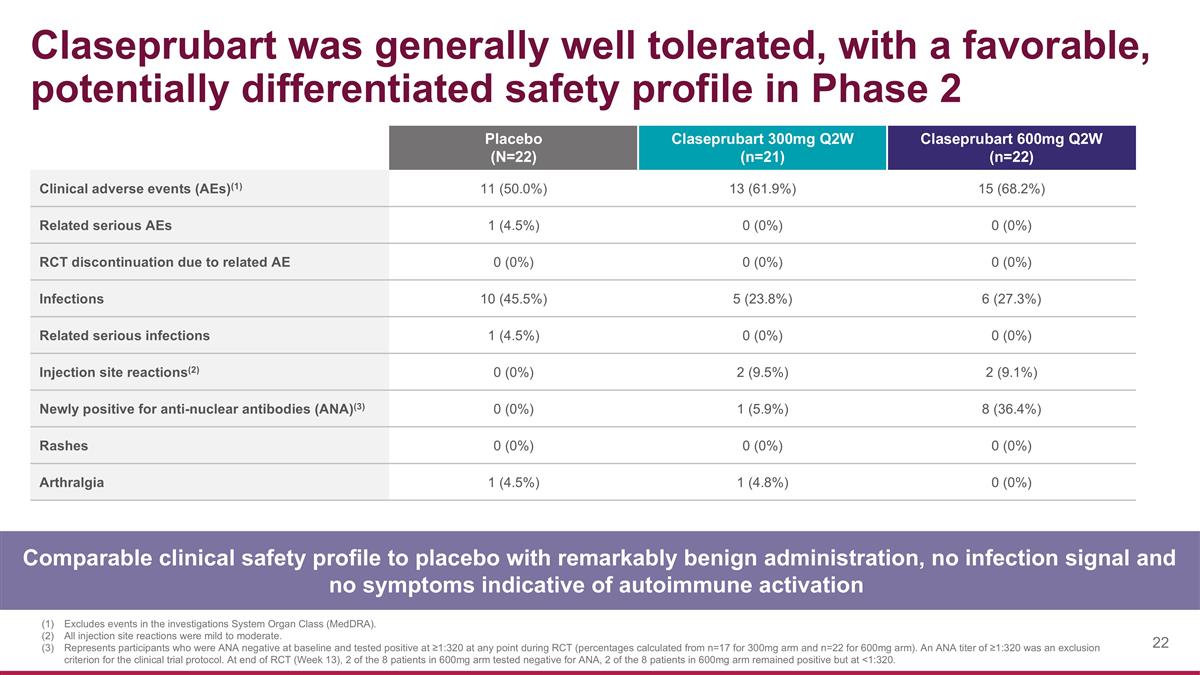
Claseprubart was generally well tolerated, with a favorable, potentially differentiated safety profile in Phase 2 Comparable clinical safety profile to placebo with remarkably benign administration, no infection signal and no symptoms indicative of autoimmune activation Placebo (N=22) Claseprubart 300mg Q2W (n=21) Claseprubart 600mg Q2W (n=22) Clinical adverse events (AEs)(1) 11 (50.0%) 13 (61.9%) 15 (68.2%) Related serious AEs 1 (4.5%) 0 (0%) 0 (0%) RCT discontinuation due to related AE 0 (0%) 0 (0%) 0 (0%) Infections 10 (45.5%) 5 (23.8%) 6 (27.3%) Related serious infections 1 (4.5%) 0 (0%) 0 (0%) Injection site reactions(2) 0 (0%) 2 (9.5%) 2 (9.1%) Newly positive for anti-nuclear antibodies (ANA)(3) 0 (0%) 1 (5.9%) 8 (36.4%) Rashes 0 (0%) 0 (0%) 0 (0%) Arthralgia 1 (4.5%) 1 (4.8%) 0 (0%) Excludes events in the investigations System Organ Class (MedDRA). All injection site reactions were mild to moderate. Represents participants who were ANA negative at baseline and tested positive at ≥1:320 at any point during RCT (percentages calculated from n=17 for 300mg arm and n=22 for 600mg arm). An ANA titer of ≥1:320 was an exclusion criterion for the clinical trial protocol. At end of RCT (Week 13), 2 of the 8 patients in 600mg arm tested negative for ANA, 2 of the 8 patients in 600mg arm remained positive but at <1:320.
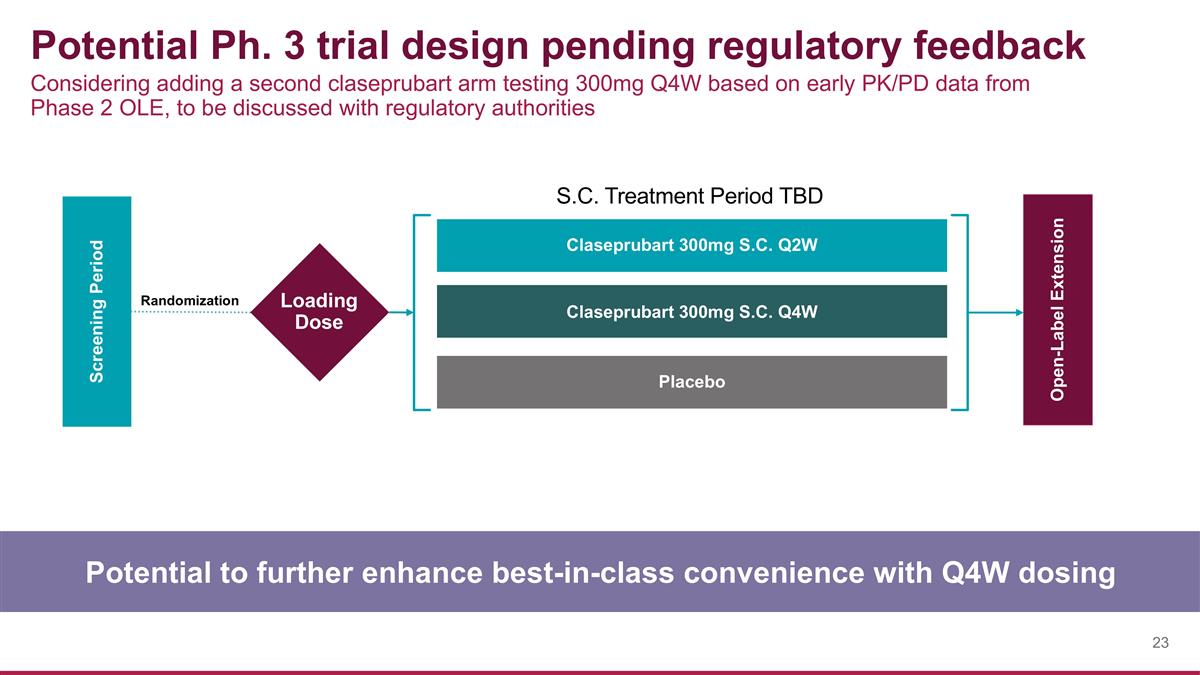
Potential Ph. 3 trial design pending regulatory feedback Potential to further enhance best-in-class convenience with Q4W dosing Screening Period Open-Label Extension Claseprubart 300mg S.C. Q2W Claseprubart 300mg S.C. Q4W Placebo S.C. Treatment Period TBD Loading Dose Randomization Considering adding a second claseprubart arm testing 300mg Q4W based on early PK/PD data from Phase 2 OLE, to be discussed with regulatory authorities

Claseprubart Summary Remarks Marino Garcia, CEO
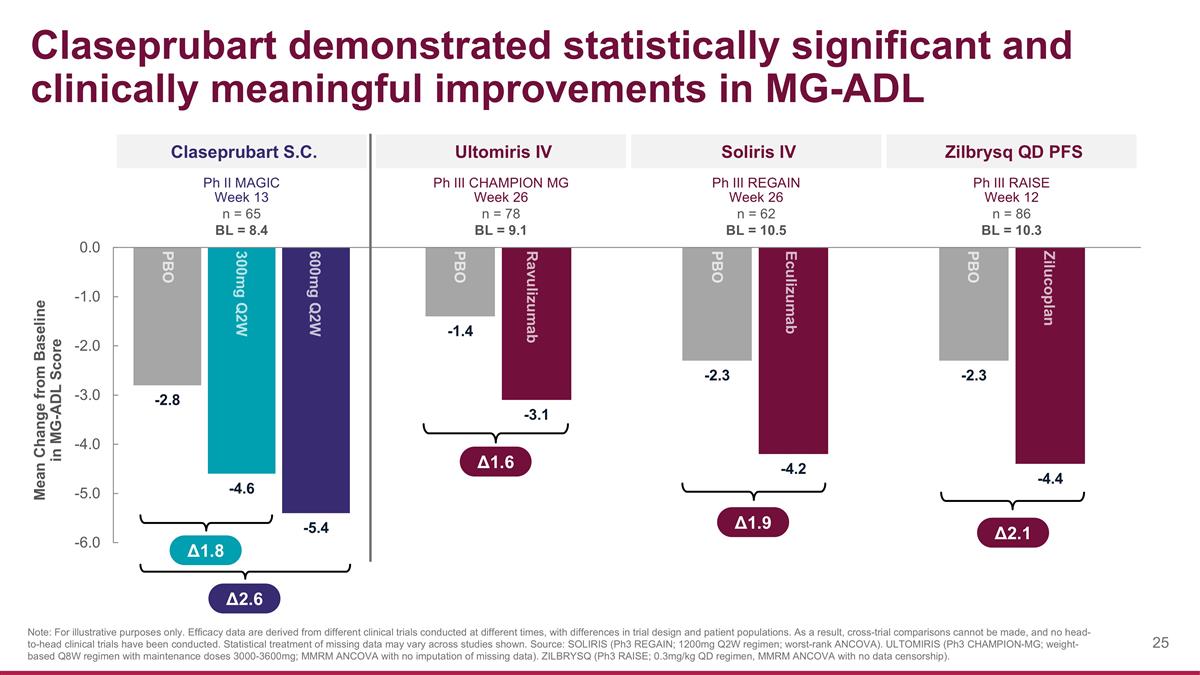
Claseprubart demonstrated statistically significant and clinically meaningful improvements in MG-ADL Claseprubart S.C. Zilbrysq QD PFS Δ1.8 Δ2.6 Note: For illustrative purposes only. Efficacy data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. Statistical treatment of missing data may vary across studies shown. Source: SOLIRIS (Ph3 REGAIN; 1200mg Q2W regimen; worst-rank ANCOVA). ULTOMIRIS (Ph3 CHAMPION-MG; weight-based Q8W regimen with maintenance doses 3000-3600mg; MMRM ANCOVA with no imputation of missing data). ZILBRYSQ (Ph3 RAISE; 0.3mg/kg QD regimen, MMRM ANCOVA with no data censorship). PBO 300mg Q2W 600mg Q2W Ph II MAGIC Week 13 n = 65 BL = 8.4 Mean Change from Baseline in MG-ADL Score Soliris IV Ph III REGAIN Week 26 n = 62 BL = 10.5 Ultomiris IV Ph III CHAMPION MG Week 26 n = 78 BL = 9.1 Ph III RAISE Week 12 n = 86 BL = 10.3 Eculizumab PBO Ravulizumab PBO Zilucoplan PBO Δ1.9 Δ1.6 Δ2.1
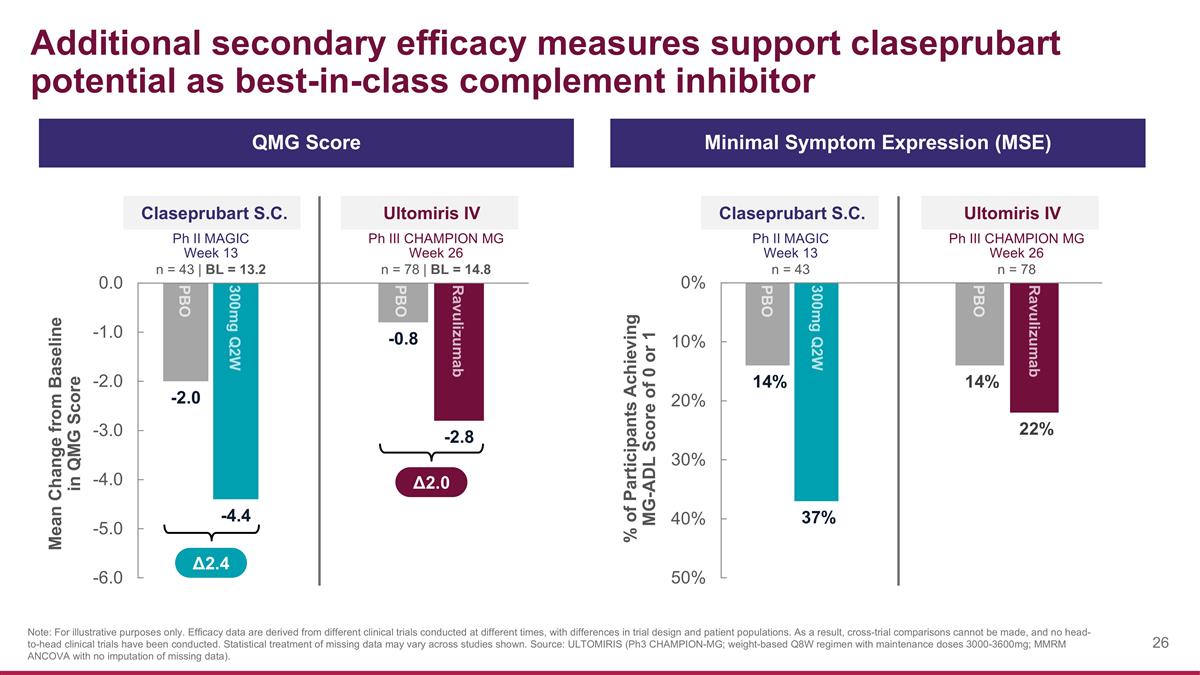
Additional secondary efficacy measures support claseprubart potential as best-in-class complement inhibitor Claseprubart S.C. Ph II MAGIC Week 13 n = 43 | BL = 13.2 Mean Change from Baseline in QMG Score Ultomiris IV Ph III CHAMPION MG Week 26 n = 78 | BL = 14.8 % of Participants Achieving MG-ADL Score of 0 or 1 Claseprubart S.C. Ph II MAGIC Week 13 n = 43 Ultomiris IV Ph III CHAMPION MG Week 26 n = 78 QMG Score Minimal Symptom Expression (MSE) Δ2.4 Note: For illustrative purposes only. Efficacy data are derived from different clinical trials conducted at different times, with differences in trial design and patient populations. As a result, cross-trial comparisons cannot be made, and no head-to-head clinical trials have been conducted. Statistical treatment of missing data may vary across studies shown. Source: ULTOMIRIS (Ph3 CHAMPION-MG; weight-based Q8W regimen with maintenance doses 3000-3600mg; MMRM ANCOVA with no imputation of missing data). Δ2.0 PBO 300mg Q2W PBO Ravulizumab Ravulizumab PBO PBO 300mg Q2W
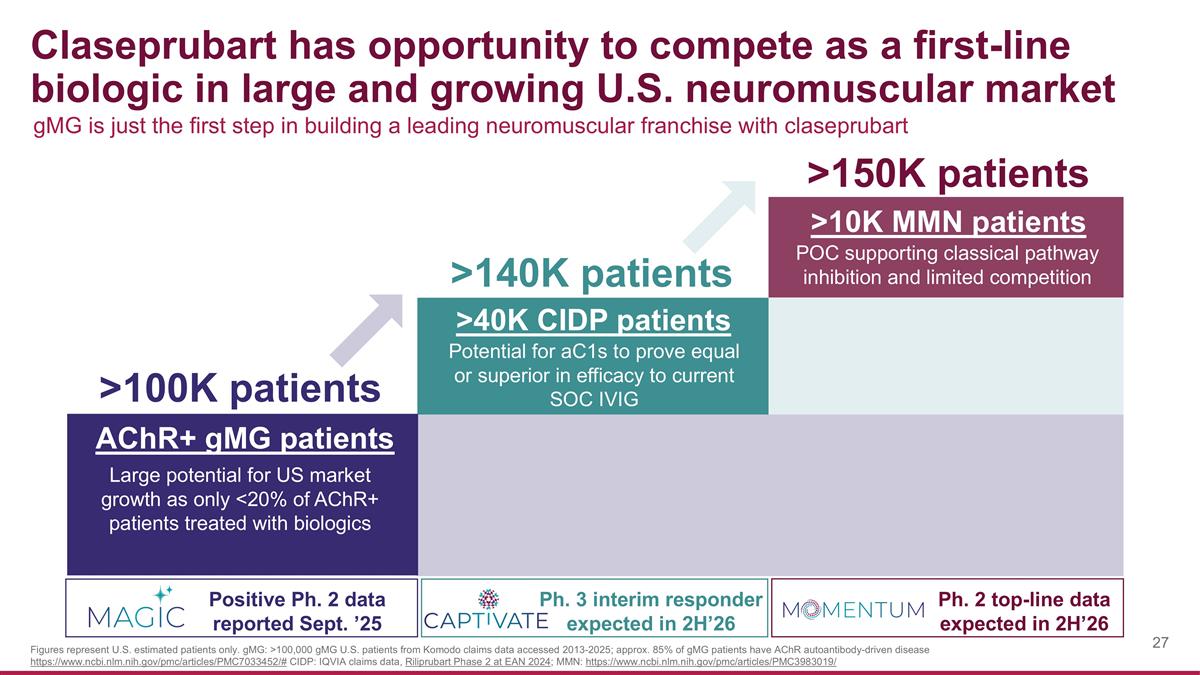
Claseprubart has opportunity to compete as a first-line biologic in large and growing U.S. neuromuscular market >100K patients >140K patients >150K patients AChR+ gMG patients Positive Ph. 2 data reported Sept. ’25 >40K CIDP patients Ph. 3 interim responder expected in 2H’26 Ph. 2 top-line data expected in 2H’26 Large potential for US market growth as only <20% of AChR+ patients treated with biologics >10K MMN patients Potential for aC1s to prove equal or superior in efficacy to current SOC IVIG POC supporting classical pathway inhibition and limited competition gMG is just the first step in building a leading neuromuscular franchise with claseprubart Figures represent U.S. estimated patients only. gMG: >100,000 gMG U.S. patients from Komodo claims data accessed 2013-2025; approx. 85% of gMG patients have AChR autoantibody-driven disease https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7033452/# CIDP: IQVIA claims data, Riliprubart Phase 2 at EAN 2024; MMN: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3983019/

Multiple internal and external near-term catalysts driven by clinical data readouts in CIDP and MMN Strong balance sheet with ~$309M(1) of cash & runway into the second half of 2027 ~35.8M shares outstanding(2) Includes unaudited cash, cash equivalents and investments as of 06/30/25 Shares outstanding on a pro forma basis, which assumes the exercise of all outstanding pre-funded warrants Based on publicly available information: https://argenx.com/content/dam/argenx-corp/media-documents/Earnings_press_release_HY.pdf.coredownload.inline.pdf Based on publicly available information: https://www.sanofi.com/assets/dotcom/content-app/events/quaterly-results/2025/2025-q2-2025-results/2025_07_31_Sanofi_Q2_2025_Results.pdf MMN gMG CIDP 2026:Claseprubart registrational Ph. 3 study initiation Phase 3 2H’26:Claseprubart interim responder analysis from CAPTIVATE expected 2H’26:Riliprubart Ph. 3 CIDP data from MOBILIZE (SOC-refractory)(4) 2H’26:Claseprubart top-line Ph. 2 data from MOMENTUM expected 2H’26:Riliprubart Ph. 3 CIDP data from VITALIZE (H2H vs IVIG)(4) 2H’26:Empasiprubart Ph. 3 MMN data from EMPASSION(3) Claseprubart Complement Peer
Pursuing the power of consistent control…with one-click! Targeting a best-in-class, first-line biologic treatment for neuromuscular diseases CLASSICAL CONVENIENCE Upstream Inhibition of Classical Pathway Only <10-Second Autoinjector Targeting No Boxed Warning or REMS Self-Administered At Home or On-the-Go Potential to Preserve Immune Function One-Click Every 2 or 4 Weeks claseprubart CONFIDENCE Aim for Potent, Rapid, Consistent Efficacy Broad Potential in Neuromuscular Diseases Potential for Best-in-Class Profile Autoinjector image for illustration purposes only. Autoinjector for claseprubart administration is anticipated to be SHL Medical’s Molly technology, patented or patent pending in the US, China, India, Japan, Korea, Taiwan and at the European Patent Office. Claseprubart is an investigational agent that is not approved as a therapy in any indication in any jurisdiction worldwide.
Q&A Top-line Ph. 2 MaGic Results in Generalized Myasthenia Gravis September 8, 2025

Appendix
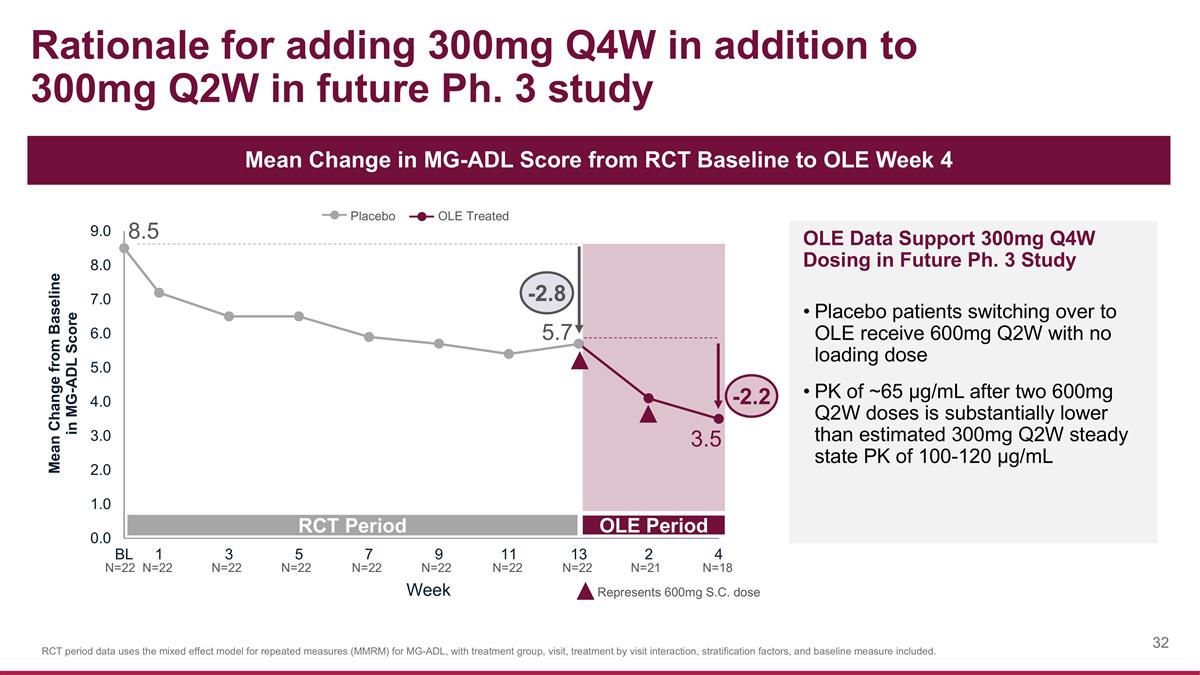
Rationale for adding 300mg Q4W in addition to 300mg Q2W in future Ph. 3 study Mean Change in MG-ADL Score from RCT Baseline to OLE Week 4 8.5 RCT Period OLE Period Placebo OLE Treated Week OLE Data Support 300mg Q4W Dosing in Future Ph. 3 Study Placebo patients switching over to OLE receive 600mg Q2W with no loading dose PK of ~65 µg/mL after two 600mg Q2W doses is substantially lower than estimated 300mg Q2W steady state PK of 100-120 µg/mL N=22 Represents 600mg S.C. dose N=22 N=22 N=22 N=22 N=22 N=22 N=22 N=18 N=21 RCT period data uses the mixed effect model for repeated measures (MMRM) for MG-ADL, with treatment group, visit, treatment by visit interaction, stratification factors, and baseline measure included. 5.7 -2.8 -2.2 3.5
