Our Mission
Developing Next-Generation Therapies for Severe Autoimmune Diseases
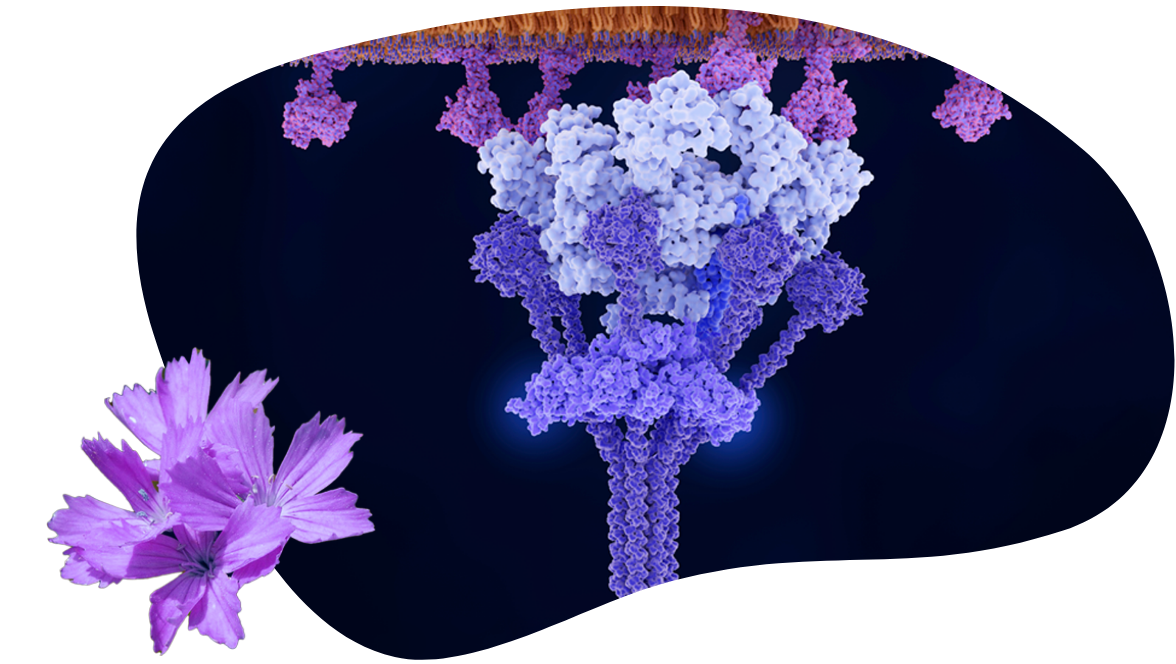
We are developing potentially best in class therapies for patients living with severe autoimmune diseases. Our lead antibody, claseprubart (DNTH103), is purposefully engineered with extended half-life, improved potency, and high selectivity for only the active C1s complement protein that drives disease pathology – enabling less frequent and more convenient self-administered subcutaneous injections. Our second clinical candidate, DNTH212 is a first and potentially best in class, bifunctional inhibitor that targets clinically validated and complementary disease modifying mechanisms, Type 1 IFN suppression and B cell modulation – enabling potential for improved clinical outcomes and patient friendly convenient, self-administered subcutaneous injections.